GS1-compliant master data automatically delivered to GDSN-certified data pools provide synchronization and mapping capabilities for data safety and accuracy. Integrated to identify and locate faulty data and product line mapping.
The SAP NetWeaver MDM GDS displays GDSN relevant data fields, like attributes and code lists, eliminating manual tasks by automatic fill for data fields and automatic publication of validated article data, transmitting master data directly from SAP as well as non-SAP solutions or Excel.
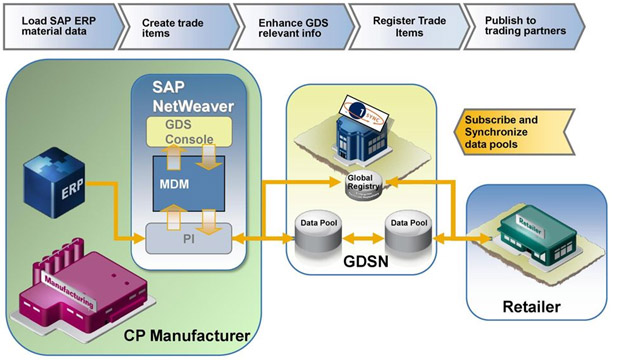
The Unovaetek can guide to choose premier comprehensive tool for data syndication, helping them to send GS1 compliant material master data directly from their own SAP and Non-SAP ERP system to GDSN-certified data pools such as 1WorldSync. GDSN plausibility checks, like data pool or food regulation requirements, are integrated part of SAP solution
GDSN message choreography is included in GDS portal, the publication or communication of product data (CIP, CIN) and feedback from data pools and trade partners (CIC) is processed automatically and documented. Automatic messaging allows users to see its target market or which GLN recipients obtained which information or data – and when the data was received. Faulty data can be corrected and resubmitted when necessary.
Unovaetek processes help standardize product data for global market share expansion via the Global Data Synchronization Network. We get you up and running with the right tools to simplify data pool connectivity.
Unovaetek understands universal best practices for product data, image capture, product data inspections and data governance – as provided by the Global Data Synchronization Network (GDSN). These best practices empower collaboration and help manufacturers attain comprehensive benefits through accurate, quality data.
Producers in the food and beverage industry face fierce competition – shrinking profit margins, rapidly changing food trends and rapidly changing consumer demands increase pressures to get the right product, with the right price in the hands of the right consumer at the right time to achieve sustainable growth.

Industry leaders understand that the producer with the best, most accurate and complete data, wins.
Clear communication and collaboration between Unovaetek, manufacturers, data recipients and the GDSN – regardless of size, sector, product type, channel or supply chain role – is essential for successful international product distribution.
GDSN quality assurance teams validate product information for completeness and accuracy – incomplete or inaccurate information is flagged for rejection, guaranteeing that when your product reaches the marketplace, it is of the highest quality and ready for purchase.
Visibility, transparency and traceability are available throughout the GDSN validation process, should any issues arise.
Unovaetek delivers the education and tools you need to simplify the process of connecting to supplier’s product data pools quickly and easily – and enhances your ability to share the information with your clients by publishing online via downloadable specifications sheets. This saves time, money and resources as accurate, consistent data is delivered from a single source.
The Unique Device Identification (UDI) system mandated by the FDA is designed to protect patient safety, improve patient safety and traceability by establishing processes to adequately identify medical devices through their distribution and inclusion of all relevant components in the GUDID registry. However, this presents a major challenge for manufacturers that lack the ability to effectively centralize and manage product data required by regulatory agencies. It is even more challenging for global manufacturers that need to submit UDI data to a myriad of country-specific regulatory agencies, each with their own unique requirements.
Innovit UDI Compliance solution is the gold standard software solution that you can use right now to comply quickly and efficiently, and their team of experts can assist you at each step along the way by helping you to stay within compliance of all the established FDA deadlines and regulatory requirements. Our solution solves these challenges by allowing device manufacturers to implement a single “global system and unified process” to aggregate, validate and submit product data to country specific regulatory agencies. Innovit’s UDI solution is also GAMP validated and 21 CFR Part 11 compliant to meet the unique validation requirements of the healthcare and medical device industry.